Problem
Every year, flu and cold season inflicts serious physical and economic damage. During the last flu season, for example, there were 36 million infections, resulting in almost 500,000 hospitalizations and 34,000 deaths, according to the CDC. And the cost to our economy was devastating, with $17 billion lost in productivity (Challenger, Gray, and Christmas, Inc).
For anyone trying to stay healthy during flu and cold season, there has been no real solution. Over-the-counter remedies are often nothing more than expensive vitamin boosts. Meanwhile, the flu shot is only partially effective, and flu drugs like Tamiflu only reduce a 7 day flu episode by less than 7 hours. These drugs can also be dangerous for individuals with compromised immune systems, children, the elderly, and those who are frail or pregnant.

Solution
Enter Vymune. Vymune is a clinically proven broad-spectrum antiviral that’s been shown to beat industry-leading treatments in the suppression of influenza. Vymune is also non-strain specific — meaning that regardless of whether you’ve caught the flu, a cold, or any other common virus, Vymune will quickly activate your immune system against the illness. Other than regulating several viruses, Vymune also provides extraordinary immune support for anyone seeking to defend their health. This sets Vymune apart from single-spectrum antiviral drugs which can cause immune deterioration when used.
Formulated with all-natural amino-acids, Vymune begins working immediately to support vigorous respiratory and immune health.
Product
Vymune is a patented, natural broad-spectrum antiviral composed of amino acids, bioflavonoids, and co-factors. Our supplement will be sold as convenient, non-prescription berry-flavored melt-away tablets, and is safe for all consumers, including the elderly, children, pregnant women, and immunosuppressed individuals. Vymune starts working immediately and is taken at the first sign of symptoms and used until symptoms stop.

To prove Vymune’s safety and efficacy, Vymedic has conducted numerous laboratory tests and two clinical studies, partnering with national, university, and government laboratories over the past ten years. Vymune has been shown to be capable of stopping 24-hour bugs, colds, and the flu. Our antiviral has also been proven to beat an industry-leading drug in the suppression of influenza.
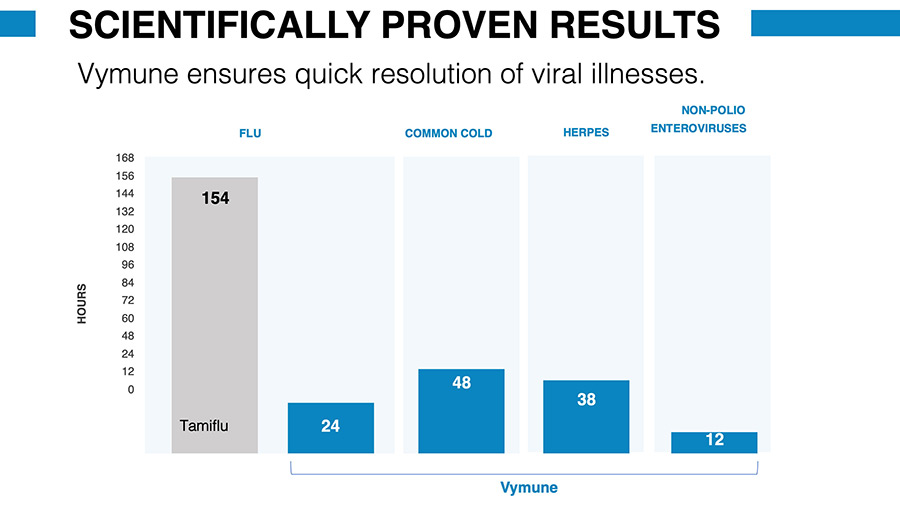
Market
The over-the-counter cough & cold medicine industry is expected to generate $10.5 billion in revenues in 2020, per IBISWorld research. Demand has never been higher for immune support and is expected to grow as consumers seek ways to mitigate viral ailments, including the flu.
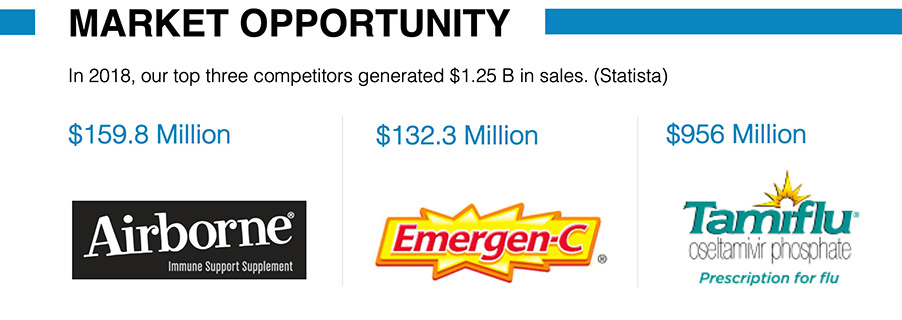
In 2018, three competing brands (Airborne, Emergen-C, and Tamiflu) sold $1.25B in the wellness space (Statista). Since none are non-strain specific and two are known to be immune suppressants, it is expected that Vymune will cause market disruption with its entry into the retail space as an over the counter broad- spectrum antiviral supplement.
Business Model
We plan to capitalize on Vymune’s ability to quickly activate the immune system safely and effectively by initially targeting consumers through Amazon.com before moving to national brick and mortar retailers, first in the United States and then expanding to Canada.
Vymune is a high-margin product, with a unit cost of $6 and a planned selling price of $20 per unit, with the ability to further reduce costs as we scale.
We will also specifically target the following customers, who may benefit from a safe, effective, immune supplement:
- Parents
- Child caregivers
- Expectant mothers
- Individuals with weakened immune systems
- Elderly individuals
- Business travelers
- Health care workers
- Workers who are exposed to the public on a frequent basis
- Educators and students
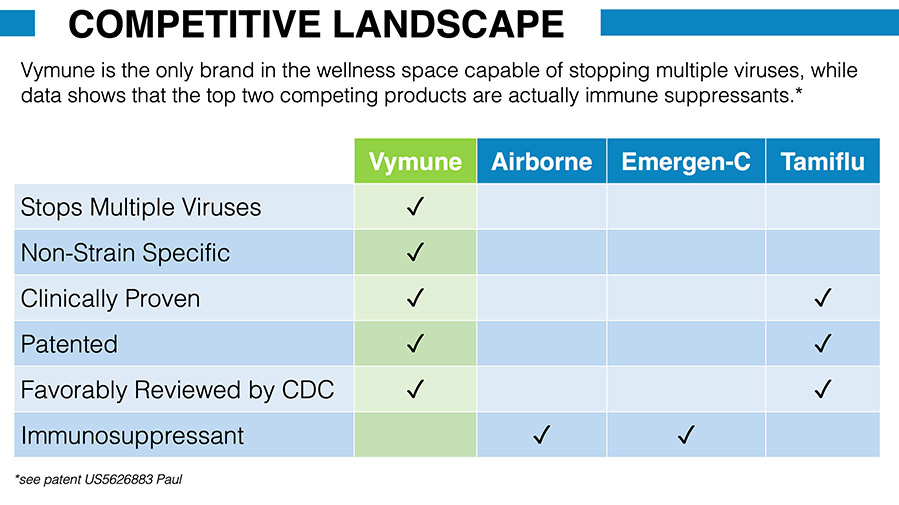
Competitive Landscape
As a broad-spectrum antiviral immune supplement, Vymune is a unique product with several competitive advantages over existing competitors, including:
- Clinically proven superior immune support
- Proven safer and more effective than competing antiviral drugs or supplements
- Stops viruses instead of just treating the symptoms
- Non-strain specific
- Favorably reviewed by CDC
- Inexpensive to produce
- Easy to use rapid melt-away tablet
- Does not require a shot, hospital, or doctor visit
Success to Date
- Successfully tested Vymune against pandemic viruses, such as H1N1, H3N2, and HSV-1
- Proven to be a safe and effective broad-spectrum antiviral in a gold-standard, randomized, double-blind, placebo-controlled human clinical trial
- Granted 24 patents

Go to Market Strategy
We are collaborating with Ogilvy, an award-winning global marketing agency, to develop a six month custom product rollout for Vymune that includes:
- Working with Ogilvy experts to design Vymune packaging
- Honing in on Vymune’s messaging and visual identity
- Creating a comprehensive media strategy
- Building and launching Amazon and direct-to-consumer channels
- Generating creative content for both direct-to-consumer and Amazon ads
Known for brand strategy, customer engagement, and ecommerce expertise, Ogilvy’s other clients include many high-profile consumer brands, including Dove, SC Johnson, IBM, Nestlé and Taskrabbit.
Team
Vymedic’s leadership team is comprised of accomplished veterans with experience in launching both products and startups. We also have a well-rounded scientific and medical team with researchers and scientists from leading institutions.
Ms. Winning has a 30-year career as a fortune 100 Product Development and Marketing Executive in two regulated industries. For the past decade, Ms. Winning has focused on the research and development of Vymune. Ms. Winning is the single largest investor in the Company. Prior to Vymune, Ms. Winning implemented marketing and design of complex technologies and networks for direct to consumer product launches. Ms. Winning was Vice President/Director of Marketing for Citicorp Retail Services, where she created innovative, credit products and global production initiatives. Ms Winning was Group Vice President of Marketing for a major MSO where she helped develop and launch networks and high tech communication services.
Steve Lehman is the Executive Chairman and a Board Member of Vymedic, Inc.. Mr. Lehman has an extensive financial and business background. Steve has been CEO of both Nasdaq and NYSE Companies. Mr. Lehman is a successful entrepreneur and investor, and was a first round investor in Mark Cuban’s broadcast.com. Mr. Lehman was an Advisory Board member of DocuSign, and is a current Board Member of Global Mentor Networks, founded by Keith Krach, Under Secretary of the US Dept. of State, for Economic Growth.
Lee M. Peterson, CPA is in private practice and has worked in public accounting since 1993. He provides income tax, accounting and financial consulting services to individuals, businesses and non-profits as well as outsourced CFO services. Prior to venturing on his own, he was a tax manager for a top multi-national CPA firm. Mr. Peterson holds a Bachelor of Science degree in Economics from California State Polytechnic University.
David E. Bartlett, Attorney-at-Law, is currently in private practice representing enterprises at the start-up, development and growth stages as outside general counsel, and on special projects, including strategic alliances, corporate and securities, mergers and acquisitions, commercial, technology, licensing and intellectual property transactions. Mr. Bartlett has co-founded several high-tech companies, and was previously a partner at Cooley Godward LLP, a Silicon Valley-based law firm. Mr. Bartlett has also authored books and articles, and is an elected member of the American Law Institute.
Scientific and Medical Team
Dr. Myron Levin, MD
Professor - Infectious Diseases
University of Colorado School of Medicine
MD, Harvard Medical SchoolGregg Solar, Ph.D.
Virology
University of Colorado
Rhodes Scholar, Oxford UniversityHeather Kroona, Ph.D.
Medicinal Chemistry
University of Minnesota
Post-Doctoral - University of MichiganKenneth Phillips, DO
Board Certified Emergency Medicine
OSU Medical SchoolLayne Los, Ph.D.
Pharmacology and Toxicology
University of Sciences, Philadelphia
Cornell UniversityCheryl Hite, MS
Immunology
University of Colorado
Use of Proceeds
If the offering's maximum Reg CF allocation of $1,070,000 is raised:
| Use | Value | % of Proceeds |
|---|---|---|
| Hiring | $119,595 | 11.2% |
| Manufacturing | $300,951 | 28.1% |
| Marketing | $597,024 | 55.8% |
| Intermediary fees | $52,430 | 4.9% |
If the offering's maximum amount of $3,000,000 across Reg. CF and Reg. D is raised:
| Use | Value | % of Proceeds |
|---|---|---|
| Hiring | $335,000 | 11.2% |
| Manufacturing | $843,000 | 28.1% |
| Marketing | $1,675,000 | 55.8% |
| Intermediary fees | $147,000 | 4.9% |
Terms
This is a side-by-side offering of Common Stock, under registration exemptions 4(a)(6) and 506(c), in Vymedic Inc.. Up to $1,070,000 may be raised under the 4(a)(6) exemption. Netcapital will determine which exemption applies to your investment and notify you before you complete your investment.
The amount raised under the two exemptions must total at least $10,000 by March 24, 2021 at 12:59pm ET. If the total doesn’t reach its target, then your money will be refunded. Vymedic may issue additional securities to raise up to $3,000,000, the offering’s maximum.
If the side-by-side offering is successful at raising the maximum amount, then the company’s implied valuation after the offering (sometimes called its post-money valuation) will be:
Pitch Deck
Financials
These financial statements have been reviewed by an independent Certified Public Accountant.
SEC Filings
The Offering Statement is a formal description of the company and this transaction. It’s filed with the SEC to comply with the requirements of exemptions 4(a)(6) and 506(c) of the Securities Act of 1933. Similar information is sometimes offered in a Private Placement Memorandum for 506(c) offerings.
All SEC filings related to this side-by-side offering are available here:
Understand the Risks
Be sure to understand the risks of this type of investment. No regulatory body (not the SEC, not any state regulator) has passed upon the merits of or given its approval to the securities, the terms of the offering, or the accuracy or completeness of any offering materials or information posted herein. That’s typical for Regulation CF or Regulation D offerings like this one.
Neither Netcapital nor any of its directors, officers, employees, representatives, affiliates, or agents shall have any liability whatsoever arising from any error or incompleteness of fact or opinion in, or lack of care in the preparation or publication of, the materials and communication herein or the terms or valuation of any securities offering.
The information contained herein includes forward-looking statements. These statements relate to future events or to future financial performance, and involve known and unknown risks, uncertainties, and other factors, that may cause actual results to be materially different from any future results, levels of activity, performance, or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties, and other factors, which are, in some cases, beyond the company’s control and which could, and likely will, materially affect actual results, levels of activity, performance, or achievements. Any forward-looking statement reflects the current views with respect to future events and is subject to these and other risks, uncertainties, and assumptions relating to operations, results of operations, growth strategy, and liquidity. No obligation exists to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, even if new information becomes available in the future.
More Info
Updates
- Aug 29, 2021Hello Netcapital Investors! Vymune is LIVE on...
- Aug 20, 2021To our valued Netcapital Investors, Our...
- Mar 24, 2021Primary offering finalized, selling shares
- Oct 30, 2020We are happy to announce that Vymedic has...
Ask a Question
Proofread your comment before submitting: once it's posted, you can’t edit or delete it. Investors are advised to review our Discussion Board Policy before submitting a comment. For the fastest help with the web site, email help@netcapital.com instead of commenting.
