Introduction
Clean Bite™ is building an advanced single use toothbrush. Made of a foodstuff, it fully dissolves and is swallowed after 60 to 90 seconds of utility. It gently cleans the users’ teeth and gums simply by chewing it, without need for water, paste or a brush.
Because it dissolves and is swallowed, Clean Bite™ can also serve as a delivery system for active pharmaceutical ingredients (API), including the delivery of vitamins, therapeutics, medications and encapsulated vaccines throughout the developing world.
Problem
The problem of poor oral hygiene affects many different aspects of society. Some of the most vulnerable populations amongst us have access to meals, but no access to oral hygiene equipment. Clean Bite, LLC aims to address these markets.
Children
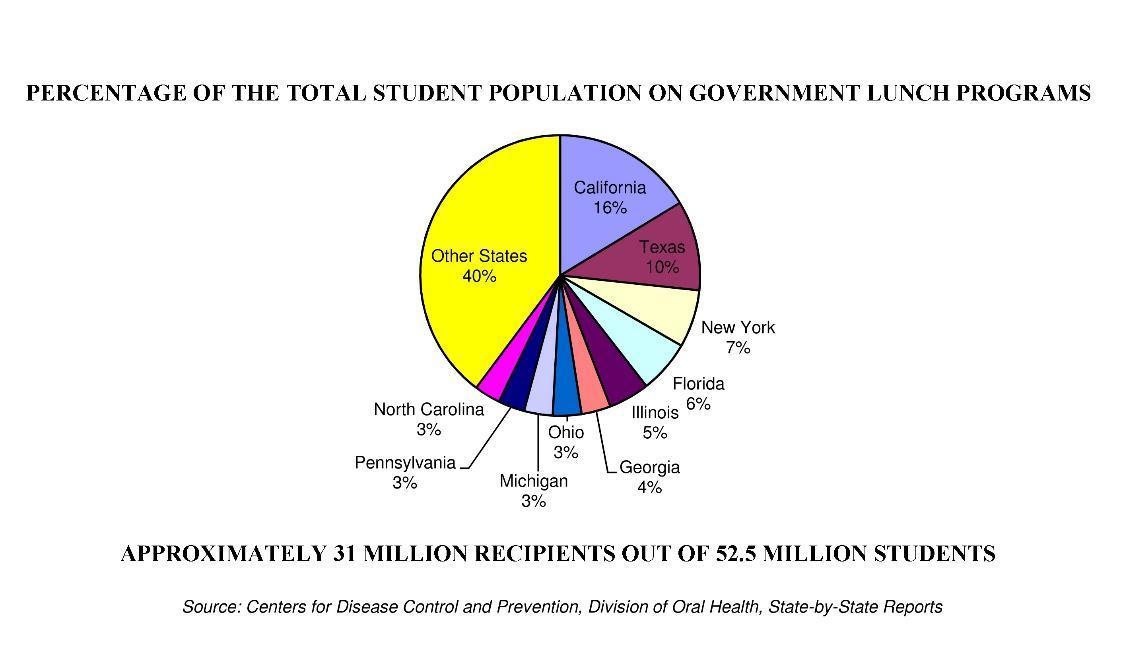
At-risk-children in the U.S. (living below, at, or near the poverty level) have two to three times the number of untreated cavities (caries) compared to their peers. These same children are the recipients of more than eight billion meals per school year and though both the CDC is aware of the problem and the USDA pays for these meals, there is no proactive measure to directly address oral hygiene on the tray.
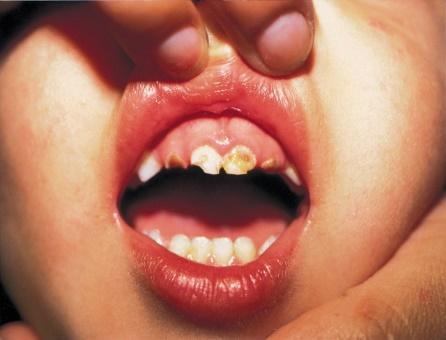
Clean Bite, LLC sees this market as one that remains completely unserved, due in part, to there not being a product that can effectively address the issues of efficacy, economy and ease of deployment.
Military and Disaster victims
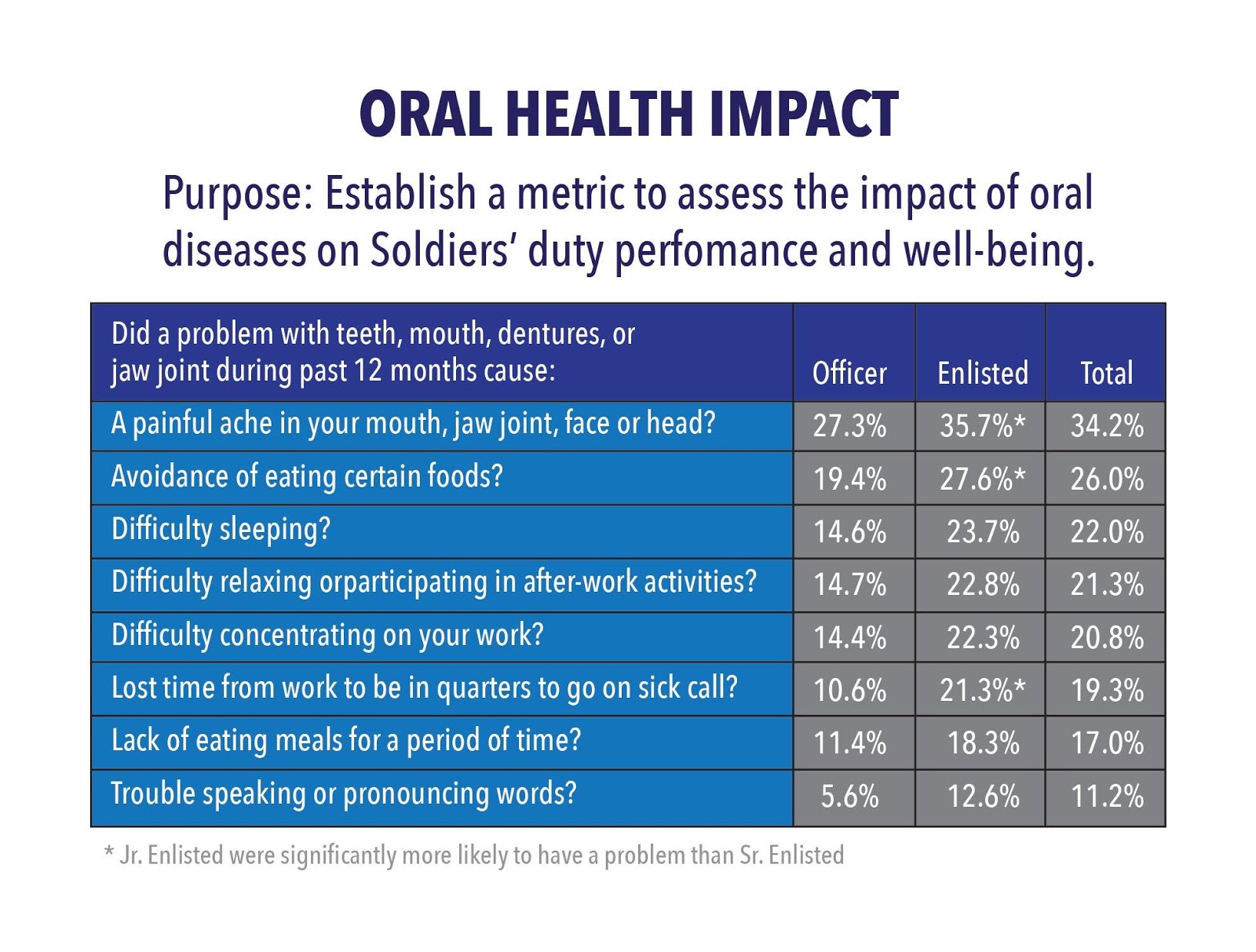
Deployed service members and victims of disaster supported by FEMA are both provided complete meals ready to eat (MRE’s). These are comprehensive meals, but the only thing provided for oral hygiene are two pellets of gum containing Xylitol. This is one more illustration where Clean Bite™ is perfectly suited to fill the gap where traditional brushing is perhaps inappropriate or unavailable.
Institutional users
- The infirm and those who rely on caregivers
- The prison system where the use of Clean Bite™ would also eliminate possible contraband
- Hospital patients acting independently without getting out of their bed
- On-the-Go public with an emphasis on travelers, air passengers, and anyone who would like to brush their teeth anywhere at any time throughout the day.
The Developing World
In the developing world, using Clean Bite™ as a delivery system for active pharmaceutical ingredients (API’s) creates the opportunity to treat various diseases, while also establishing a daily oral health regimen by the world’s poorest populations. This is not an area of expertise for Clean Bite, LLC, and Clean Bite’s use in this arena will call for strategic partners in the pharmaceutical industry.
Solution
Clean Bite™ plans to build a better single-use toothbrush.
- Shaped like a mouth piece
- 800 bristles throughout the bite channels and across the palate arch
- Four reservoirs of dentifrice with xylitol
- Made from gelatin and contains 5g of protein
- Has 60 to 90 seconds of utility, then dissolves and is swallowed
- Has different flavors and comes in adult and children’s sizes
- Economical
- Capable of acting as a delivery system for vitamins, therapeutics, medications and encapsulated vaccines, referred to as API’s (Active Pharmaceutical Ingredients)
It was designed by an Army medic while working on an amputee ward, to specifically accommodate soldiers who had lost the use of their arms in war. These patients needed a device that could brush the teeth then dissolve and be swallowed so there was no need for disposal or rinsing with water.
Despite its start as a device for wounded soldiers, the team at Clean Bite, LLC realized that there were many other populations that would benefit from a single use oral-health solution.
Statistics reveal the need to intervene proactively to better the oral health for at-risk-children. The most applicable time is when they are receiving meals. Clean Bite™ may help to prevent cavities, is cost neutral due to its protein content, and will be instrumental in forming a life-long oral health habit. Better overall health going into adulthood would reduce the healthcare costs to the government and insurance companies, while improving the health of our nation’s future aging population.
Market
Primary Market Potential
Annual Units
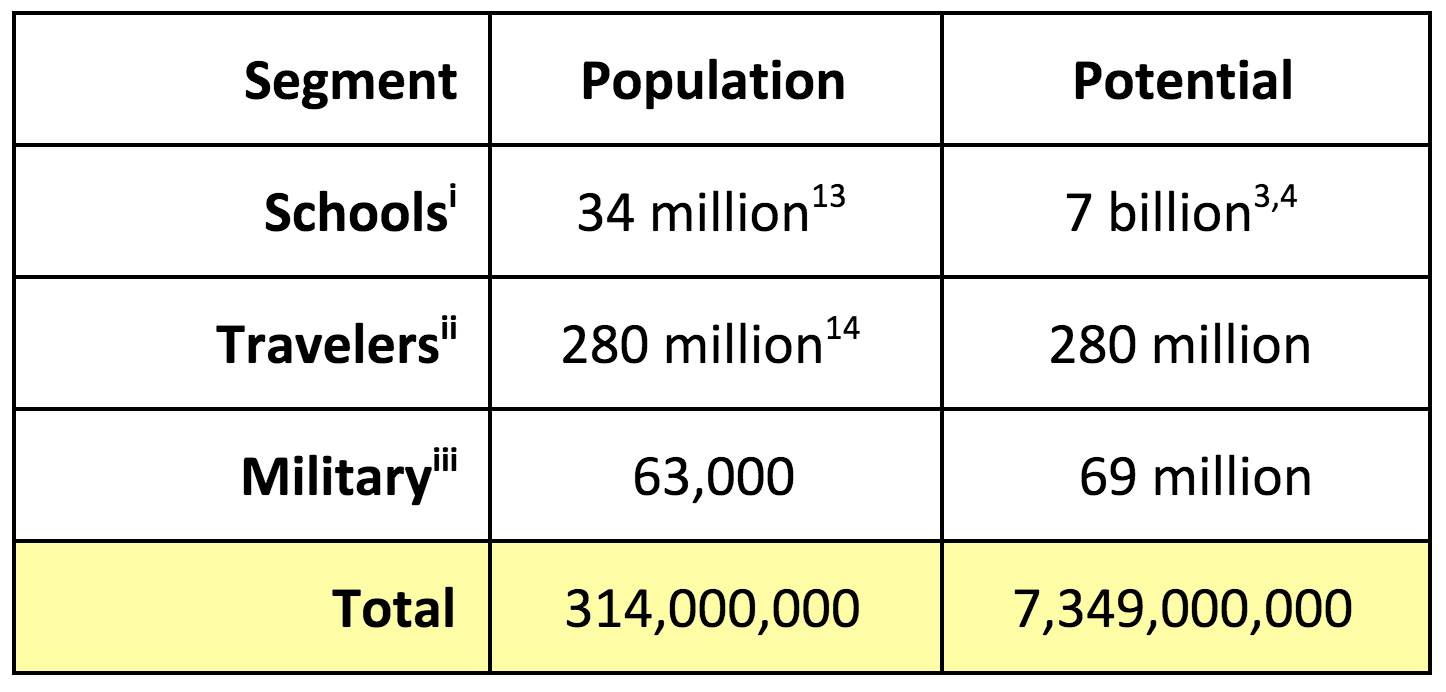
- Potential based on single unit use per day by 180 days
- Potential based on single unit use per year
- Potential based on three MRE’s per day by 365 days per year
Primary Market: U.S. Dental Hygiene

- School Students: The USDA provides ≈ 1.5 billion meals per school year to low income families. An additional 5.5 billion lunches through similar programs administered by individual school districts. These children are two to three times more likely to have untreated than that of their peers. Two US Surgeon General's reported untreated caries as the most prevalent, and preventable disease among children, yet nothing has been done to intervene when meals are served. Each Clean Bite™ contains 5 to 6 grams of protein inherent to the collagen and at a cost of 33¢, the price is competitive with most every protein bar on the market. Clean Bite™ then becomes a two-for-one, delivering oral hygiene and protein for the price of one.
- Military: “Deployed” troops are provided three (3) MRE’s (Meals Ready to Eat) a day. Included are two (2) pellets with Xylitol to restore the level of pH and freshen breath. This insufficiency in delivering oral hygiene contributes to the 17% of all combat troops need evacuation from the theater for dental emergency, based on a one-year tour.
- Travelers: Some 100 million Americans board domestic airlines an estimated 740 million times annually. Roughly, 35% of these passengers, or 260 million, are flying between 7:00 P.M. and 7:00 A.M. “normal brushing times.” Additionally, there are 3.5 million long haul truckers who spend, on average, 50 weeks a year on the road.
Secondary Markets
The unique design and functions of the Clean Bite™ should ultimately open the door to a vast overseas market for delivering vitamins, various medicines, nutrients, therapeutics, and potentially microencapsulated vaccines. Clean Bite™ could contain virtually any nutrient, therapeutic compound, or microencapsulated API. The full promise of the Clean Bite™ technology should, in the future, be exploited to the benefit of the world’s poorest populations where disease and malnutrition are rampant.
Additional Sub-markets:
- Hospitals: The national average of daily inpatients numbered 592,000 in 2013, with an average stay of just under five days
- Prison Inmates: The U.S. DOJ in 2015 recorded the number, nationwide at 2.2 million
- Campers: There are almost 14 million adult campers who spend an estimated two to six nights annually in our National or State parks.
- International Oral Hygiene: The potential population for our purpose is set at five (5) billion people, and market size is set at 5B x 365 ≥ 1.8T, with potential brushings being of a fractional percentage of use through each year from 2018 to 2027. (NB: not calculated)
- International Oral Hygiene and Delivery System: Estimated as being one (1) billion people (conservatively) that may live in circumstances where the Clean Bite, may be applicable as a viable delivery system. The market size ≈2B people and use is as above noted. (NB: not calculated)
Competitive Landscape

Business Model
Clean Bite, LLC plans to use its expertise to develop high-speed endothermic injection equipment to manufacture the patented Clean Bite™. At the completion of this milestone, Clean Bite, LLC plans to open a small plant with the capability to produce approximately 15M units per year. This facility should act as a control center to hone every aspect of Clean Bite’s production from equipment utilization and maintenance, integration of two shifts, plant layout, packaging and shipping. It should also serve to provide finished product for testing for efficacy, focus groups, samples and some regional sales.
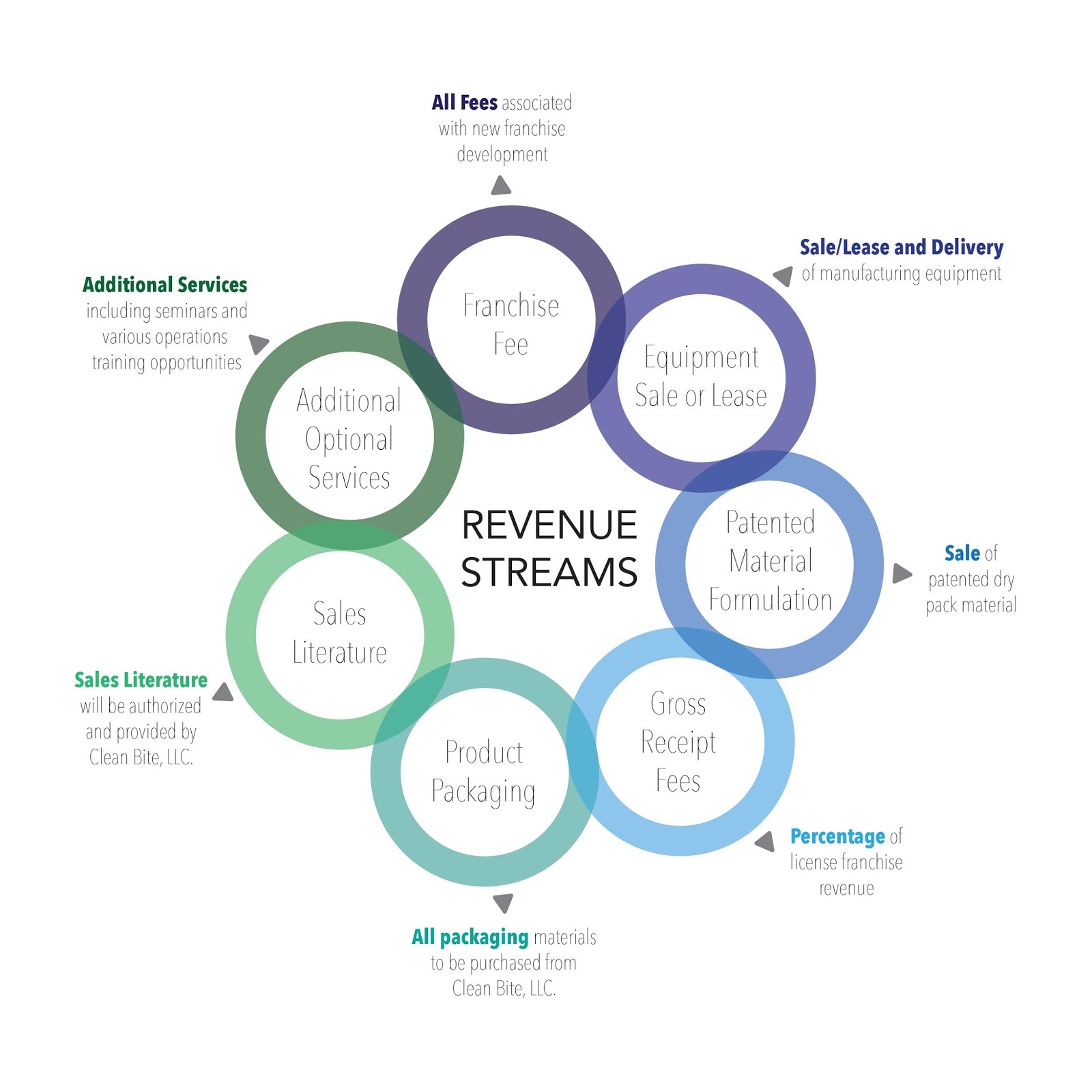
The Business Model will then be put into operation to establish a national Licensing Franchise Manufacturing company that will target multiple revenue streams consisting of:
- Franchise Fees
- Manufacturing and Packaging Equipment Sales or Leasing
- Sales of Dry Pack Formulated Material and Dentifrice used to make Clean Bite™
- Gross Receipts Fees
- Packaging Material
- Product Literature
- Other Optional Contract Services
- Equipment & Plant Maintenance Training
- New Employee Training
- Marketing & Sales Training
- Contract Sales Documents
Gross Receipts fees are based on gross sales volume when converted to the Franchisor’s Suggested National Accounts Pricing (FSNAP). Actual sale prices may vary above or below the FSNAP, at the discretion of the sub-licensee, but Gross Receipts Fees will be audited based on a minimum mandatory production, using the FSNAP, and correlated to the consumption of material. The purpose of establishing FSNAP is to enable all Licensed Franchise Manufacturers to participate in the supply of product to large accounts, e.g. military, FEMA, hospital, independent distributors, etc., that maybe established directly by Clean Bite, LLC.
Progress
Clean bite, LLC has three phases to go through and each has its own customer. First and most important are potential investors in Clean Bite, LLC and to acquire their support they need to know some basics about who we are and what Clean Bite™ is, and its potential part to play in expanding oral hygiene.
We plan on our second-round of customers also being investors, the difference being that their investment will be in their own business as a Licensed Franchise Manufacturer. These investors will be part of a regional makeup of manufacturers who will provide the backbone of expansion, while reducing the overall financial commitment, by dividing the cost of what otherwise would be a single entity.
The third customer is the consumer or in some cases the purchaser, be it a government agency, NGO, or institutional entity. The adaptation toward what is known as the tipping point will be based on the attributes of Clean Bite™ that are not found in totality in any products in the oral hygiene space that augments traditional brushing when it is unavailable or inappropriate.
Each of these three categories of customers will have different reasons for being involved with Clean Bite, LLC, beginning with investors and ending with consumers.
- The first-round of initial investors will be inclined perhaps because Clean Bite™ is patent protected and unlike any other product. Emphasis however, will likely be on the business model’s use of Licensed Franchised Manufacturing to contain the cost to scale. It also helps that the global oral care market from 2018 through 2024 is projected to be 37.06 to 44.43 (in billions of U.S. Dollars).www.statista.com
- The second-round investors will be franchisors who will look at the costs of operations, new product introduction, outcomes on efficacy testing, competitive products, and the ability to be part of collectively contributing product to national account sales. The new tax law will also be favorable with accelerated expensing on the cost of capital equipment, along with market size and many other considerations. A major factor will be Clean Bite, LLC’s own plant operation that will serve as a showcase for a well-oiled machine.
- The end consumers are divided into two groups, from the occasional user to those who may be part of larger groups such as students, military, and others that are cited elsewhere throughout. For the individual use it’s a matter of personal satisfaction; for those users adapting resulting from organizational buying, it will be a matter of efficacy and measured outcome.
The statistics that bear this out are daunting and voluminous. It is true that poor oral health can lead to an overwhelming number of diseases and conditions that are life threatening. Far too many to go into here beyond what has already been discussed. This means that these first-round customers will likely be Doctors, RDH’s, Social Workers who see things as they are, some Veterans because of their own experiences and the fact that Clean Bite’s origin is rooted in military service, and finally those who are market savvy.
Success
Clean Bite™, LLC is a newly formed company, and is affiliated with Dent-Chew Brush, LLC (DCB) that was started in January 2009, as a licensing firm, with the objectives to design, patent, and prototype a single use toothbrush made from a foodstuff. The expectation was that having achieved these goals, a global consumer manufacture would License, and manufacture the IP covered by the patent.
Clean Bite™ is made of gelatin, and early on Kodak and its subsidiary Eastman Gelatine, Inc. pursued DCB as Strategic Partners wanting to be the sole supplier of gelatin globally. Kodak assisted in R&D in exchange for warrants until it sought protection under a Chapter 11 of the Bankruptcy Code. The loss of Kodak was a setback but the passage of the new JOBS Act, of April 5, 2012 had promise of investors however Title III was delayed until May 16, 2016 and flawed.
In late 3Q 2016 Johnson & Johnson requested a meeting. Their only interest was in buying a $100M firm to turn it into a $1B division while branding Clean Bite™ with the Listerine name for use in the developing world for populations that did not have reliable potable water.
This was the watershed moment for Gallagher, in changing the company’s business model from a licensing firm to a Licensed Franchise Manufacturing. Gallagher had bartered his own equity for the preceding four years to keep furthering R&D, which enabled development of the endothermic injection equipment to make Clean Bite™, so the stakes were too high not to push for change among his partners.
It took Gallagher six months to formulate a business plan that would chart a path from funding for the developing of high-speed manufacturing equipment, efficacy testing, and franchising on national level. The final plan was accepted in February 2018 after negotiating a buyout that was equitable to his twelve partners while transferring the Patent rights, all proprietary knowledge and physical assets to Clean Bite, LLC.
Press mentions:
Team
What makes this team unique is that three of its members have worked together on this endeavor for ten years under a different business model. With the change in the business comes the ability to go directly into manufacturing using all the arcane knowledge that has been gained over the past decade. Each milestone has been designed to reduce the capital expenditure, while acquiring greater value with each accomplished goal in the critical path to full franchise manufacturing and distribution.
The knowledge that was accumulated over the years and anticipated to be used in the support of a licensee, will now be utilized internally with firsthand experience.
As the inventor, he has developed the Clean Bite™ from concept to reality and built the development team. Previously, he was SVP of AmeraCash Solutions, Inc., where he oversaw the recruitment of 400 independent merchants to establish Philadelphia’s largest POS network of utility payment locations. Prior to that he founded and was President of both American Recreational Builders, Inc. which constructed athletic facilities from 1973 to 1979 and was renamed Gallagher and Associates, Inc. when merging with Gallagher and Associates (Palm Springs, CA) transitioning to a unionized construction company specializing in the restoration of concrete and steel and the application of protective coatings. Primary clientele was the U.S. government (military, federal and state agencies) and industrial plants with projects ranging from U.S. Naval ships to bridge and highway infrastructure.
Dr. Bielski is a Director of Clean Bite, LLC, and Chemist with expertise in the total synthesis of carbohydrates and pheromones, formation of micro and nanocapsules for the controlled delivery, synthesis of polymers and radio-labeled compounds, ultra-sensitive detection and measurements in biological fluids, food, and petroleum products. Dr. Bielski co-founded several small companies (Petramec, Petrotraces, Attochrom/Biotraces). At present, he is a Senior Scientist in Value Recovery, Inc., one of four partners in Cheminnolab, LLC., Adjunct Professor at the Pharmaceutical Department of the Wilkes University and has been elected a Secretary of the Chemical Consultants Network. Dr. Bielski developed a novel technology enabling the recovery of various waste components (using phase transfer catalysis), a method that economically destroys methyl bromide from fumigations and a novel material formulation for a disposable oral hygiene product based on cross-linked gelatin. Dr. Bielski holds a master’s degree in chemical engineering and a Ph.D. in organic chemistry (with O. Achmatowicz of the Achmatowicz reaction). He was a postdoctoral fellow at Imperial College in London with the Nobel laureate Sir Derek Barton. He worked in academia at Warsaw Agricultural University, Cornell University, and Lehigh University.
Thomas Holder is a Member of Clean Bite, LLC and he is responsible for the design, and assembly of the first endothermic injection equipment, and will lead in the development of the high-speed endothermic injection equipment. Mr. Holder is also the CEO of Thomas Holder Inc., a design firm he established in 1987. His CAD design expertise includes SolidWorks, the most popular 3D solid modeling software. Mr. Holder holds a BS from California State College (now California University of Pennsylvania) in Industrial Arts Education, and a Master in Industrial Arts from Millersville University. Mr. Holder has designed, developed and built food production equipment and related plant packaging lines for some of the most familiar food brands in the U.S.
Craig Callaghan, Esq. is Clean Bite, LLC's In-House General Counsel. Prior to joining DCB Craig practiced law for more than 25 years as a litigator at top-tier firms in Philadelphia. He has represented clients in business counseling, complex litigation, and alternative dispute resolution. Craig is experienced in handling internal investigations, risk management, labor and employment, trade secrets and mass torts. He has also served as an arbitrator in the Philadelphia County Court of Common Pleas Compulsory Arbitration Program and as a trained certified mediator. His clients included, among others, major pharmaceutical companies and a top five accounting firm. Craig is an honors graduate of Thurgood Marshall School of Law where he graduated top of his class. He has been certified as a trained mediator by the Lancaster Mediation Center. Craig also attended the University of Houston where he obtained his bachelor’s degree in English literature. Before attending law school, Craig taught remedial reading to 7th and 8th-grade inner-city students in the Houston Independent School District. Following law school, Craig taught at Villanova University School of Law as a legal writing instructor. Bar Admissions: Texas (Inactive), Pennsylvania, U.S. District Court, Eastern District of Pennsylvania.
Use of Proceeds
If the offering's maximum amount of $1,068,000 is raised:
| Use | Value | % of Proceeds |
|---|---|---|
| Compensation for managers | $168,240 | 15.8% |
| Contractual Licensing & Royalty Fees | $53,500 | 5.0% |
| Operating Budget | $19,526 | 1.8% |
| Marketing | $17,225 | 1.6% |
| Professional Services: Accounting & Engineering | $390,100 | 36.5% |
| Personnel: Admin Assistant & Web/Soc. Media | $21,594 | 2.0% |
| Plant Operations: 4th Q. | $10,000 | 0.9% |
| Health Insurance | $9,900 | 0.9% |
| State & Federal Employment Taxes | $12,039 | 1.1% |
| Cash Reserved for (13th Mo.) 5th Q. Equip. Purchase | $313,544 | 29.4% |
| Intermediary fees | $52,332 | 4.9% |
Terms
This number includes all funds raised by the Company in this round on Netcapital. This is an offering of Units, under registration exemption 4(a)(6), in Clean Bite, LLC. This offering must reach its target of at least $10,000 by its offering deadline of October 19, 2018 at 4:00pm ET. If this offering does not reach its target by the offering deadline, then your money will be refunded.
If the offering is successful at raising the maximum amount, then the company’s implied valuation after the offering (sometimes called its post-money valuation) will be:
Pitch Deck
Financials
These financial statements have been reviewed by an independent Certified Public Accountant.
SEC Filings
The Offering Statement is a formal description of the company and this transaction. It’s filed with the SEC to comply with the requirements of exemption 4(a)(6) of the Securities Act of 1933.
All SEC filings related to this offering are available here:
Understand the Risks
Be sure to understand the risks of this type of investment. No regulatory body (not the SEC, not any state regulator) has passed upon the merits of or given its approval to the securities, the terms of the offering, or the accuracy or completeness of any offering materials or information posted herein. That’s typical for Regulation CF offerings like this one.
Neither Netcapital nor any of its directors, officers, employees, representatives, affiliates, or agents shall have any liability whatsoever arising from any error or incompleteness of fact or opinion in, or lack of care in the preparation or publication of, the materials and communication herein or the terms or valuation of any securities offering.
The information contained herein includes forward-looking statements. These statements relate to future events or to future financial performance, and involve known and unknown risks, uncertainties, and other factors, that may cause actual results to be materially different from any future results, levels of activity, performance, or achievements expressed or implied by these forward-looking statements. You should not place undue reliance on forward-looking statements since they involve known and unknown risks, uncertainties, and other factors, which are, in some cases, beyond the company’s control and which could, and likely will, materially affect actual results, levels of activity, performance, or achievements. Any forward-looking statement reflects the current views with respect to future events and is subject to these and other risks, uncertainties, and assumptions relating to operations, results of operations, growth strategy, and liquidity. No obligation exists to publicly update or revise these forward-looking statements for any reason, or to update the reasons actual results could differ materially from those anticipated in these forward-looking statements, even if new information becomes available in the future.
More Info
Updates
- Oct 19, 2018Primary offering finalized, selling units
Ask a Question
Proofread your comment before submitting: once it's posted, you can’t edit or delete it. Investors are advised to review our Discussion Board Policy before submitting a comment. For the fastest help with the web site, email help@netcapital.com instead of commenting.



